Advancing the first meaningful oral therapeutic for people living with dystonia and related movement disorders
Our approach is intended to expand the possibilities for people living with movement disorders—to help them move freely.

Targeting the source, not just the symptoms
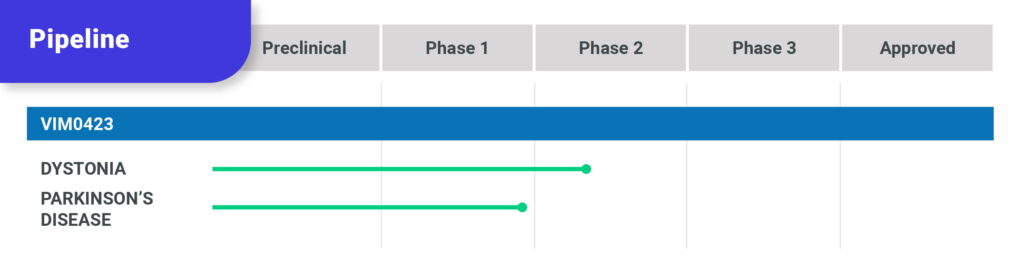
Our lead program, VIM0423 is a novel oral therapeutic for dystonia that selectively targets muscarinic cholinergic receptors in the brain. These receptors are known to be involved in dystonia and related movement disorders but targeting them has been limited by poor tolerability. VIM0423 has been designed to maximize both efficacy and tolerability to deliver effective therapy for all dystonia patients.
VIM0423 has been granted Fast Track designation by the FDA and is currently being evaluated in a Phase 2 clinical trial in patients with dystonia. Vima plans to initiate a Phase 2 trial in Parkinson’s disease in 2026.
Current drug development pipeline
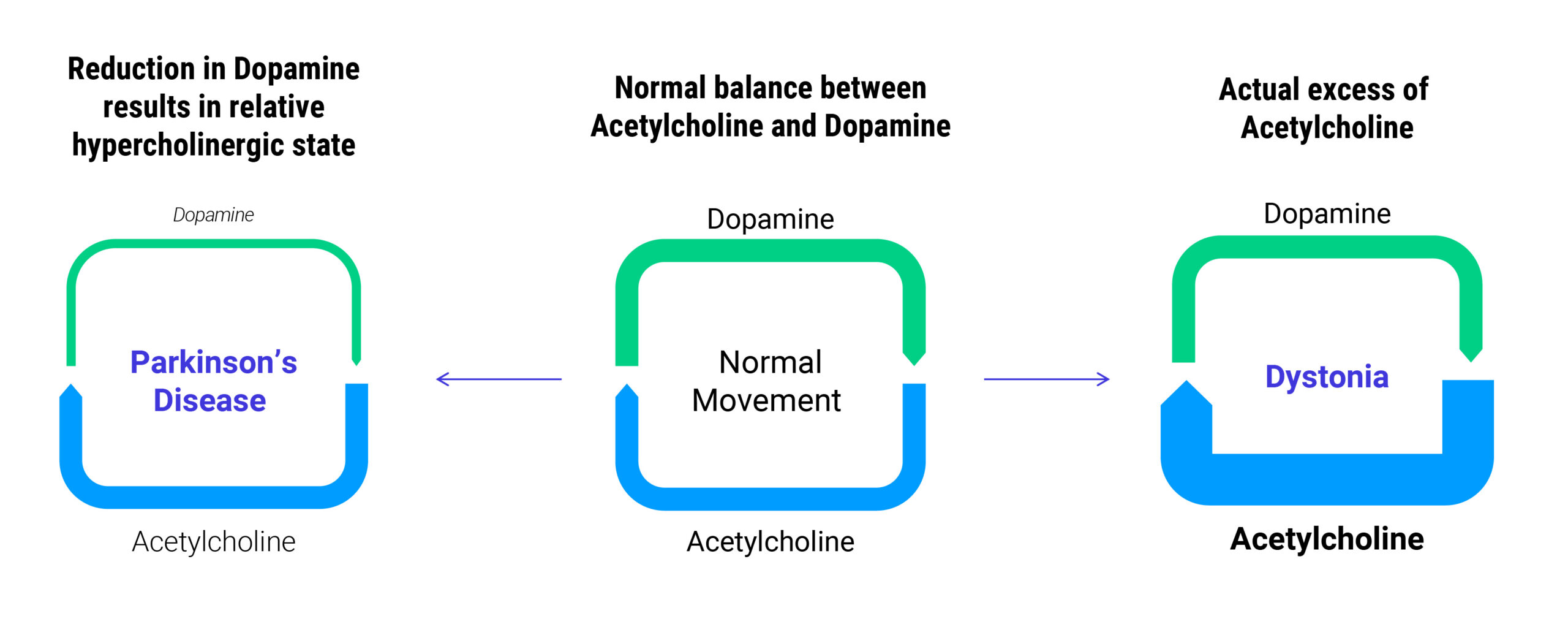
Both dystonia and Parkinson's disease are neurological conditions that disrupt motor control and can severely affect daily function
Dystonia and Parkinson’s disease have overlapping clinical symptoms and biological mechanisms. Both conditions are characterized by an imbalance between dopamine and acetylcholine in the brain, leading to heightened cholinergic activity and disruption of motor control—a key class of receptors that are fundamentally involved in the response to reduction in dopamine results in relative hypercholinergic state are the muscarinic receptors which present an important target for treating these movement disorders.
Phase 2 clinical study of VIM0423 in isolated dystonia
Stride Dystonia, our Phase 2 clinical study in isolated dystonia, is currently recruiting patients. For more information, please visit these resources for more information on the study.
News
Learn about our progress and latest milestones.